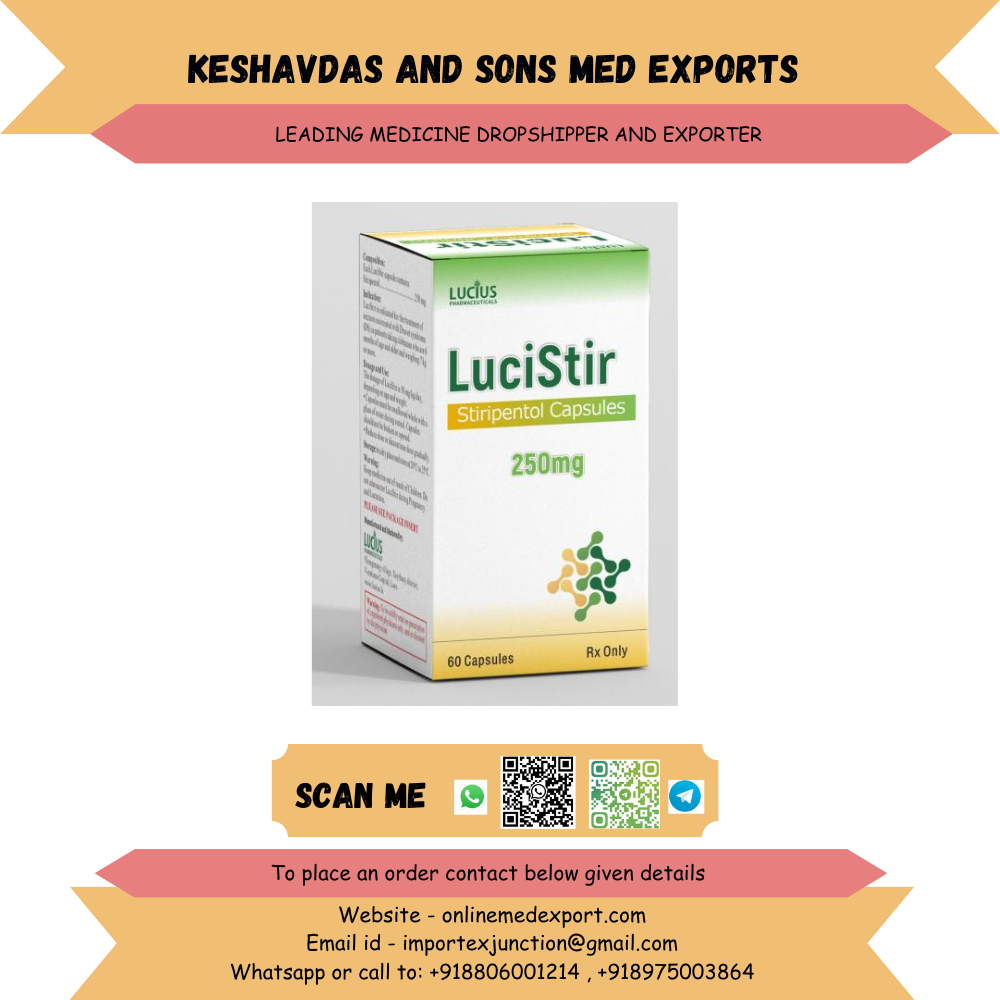
COMPOSITION: Each LuciStir capsule contains Stiripentol……………………………. 250 mg INDICATION: LuciStir is indicated for the treatment of seizures associated with Dravet syndrome (DS) in patients taking clobazam who are 6 months of age and older and weighing 7 kg or more. DOSAGE AND USE: The dosage of LuciStir is 50 mg/kg/day, depending on age and weight. • Capsules must be swallowed whole with a glass of water during a meal. Capsules should not be broken or opened. • Reduce dose or discontinue dose gradually. STORAGE: in a dry place and store at 20°C to 25°C. MANUFACTURED AND MARKETED BY: LUCIUS PHARMACEUTICALS(LAO) CO., LTD No.26, Thongmang village, Xaythany district, Vientiane Capital, Laos